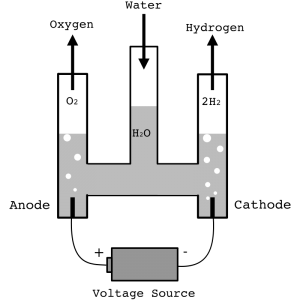
 In two posts we had a look at the background science which is necessary to consider the question of the hydrogen economy myth. Looking at the laws of thermodynamics suggests this idea does not add up. Beyond these further inherent inefficiencies make the whole concept seem even less worthwhile, even if in fuel cells the excess heat produced by the process can be captured and utilised for combined heat and power. Nevertheless when people talk about the hydrogen economy they are largely thinking about either storing electricity as hydrogen or using it as a means of transport. The reader has probably got some questions at this point and I have tried to anticipate them.
In two posts we had a look at the background science which is necessary to consider the question of the hydrogen economy myth. Looking at the laws of thermodynamics suggests this idea does not add up. Beyond these further inherent inefficiencies make the whole concept seem even less worthwhile, even if in fuel cells the excess heat produced by the process can be captured and utilised for combined heat and power. Nevertheless when people talk about the hydrogen economy they are largely thinking about either storing electricity as hydrogen or using it as a means of transport. The reader has probably got some questions at this point and I have tried to anticipate them.
Is there enough platinum?
Platinum is used both in the electrolyser and PEM fuel cells. Its main use at present is in catalytic converters in vehicles. Minute amounts are constantly lost from these and end up in our roads, the fact that research is going to use bacteria to bind to this platinum and allow its collection suggests we cannot have absolute confidence in platinum supplies. At least in theory it maybe be possible to use enzymes. Some bacteria make hydrogen. The quantity is severely limited by the laws of thermodynamics, but both electrolysis and electricity production could be possible this way. The problem would be the enzymes would degrade and would need regularly replacing. Whilst the efficiency might be a bit better than chemical systems its unlikely the overall efficiency of the whole process would make it worth pursuing.
Are there other fuel cell alternatives?
Yes there are a variety of other fuel cell technologies. Solid oxide fuel cells have the advantages that they can directly use a variety of fuels such as methane and don’t use platinum. PEM fuel cells can only use methane indirectly through the shift reaction which produces CO2 and H2 via a carbon monoxide intermediate. Carbon monoxide poisons the platinum catalyst even at very low concentrations. However solid oxide fuel cells can even use carbon monoxide as a fuel. Even so their efficiency is no better than PEM cells and they operate at very high temperatures meaning that some energy would be needed to get them started (as would PEM cells but their lower operating temperature means this would be less).
Any other problems that you see?
Yes I’m afraid there are a few. One relates to transportation of hydrogen. Compressing it takes energy and its not very energy dense, its been estimated that compared to fossil fuels if we were going to transport it for use by road for vehicle use we would require 15x as many tankers at present. Transport by pipes is more efficient but still has much larger energy losses than methane.
Another problem relates to its use in domestic fuel cells. Of the recoverable energy in such devices about 50% would be electricity and 50% heat. Currently we use a lot more heat than electricity. The average boiler is rated in the UK at somewhere in the range 10-30kWh peak output. So if you wanted a fuel cell to meet 10kWh of heat it would also produce 10kWh of electricity, far more than we need almost all the time. Such a system would also be enormous, a German company produced a domestic 5kWh fuel cell which was a the size of a chest freezer. The solution is what happens with domestic Sterling Engines on the UK market which is they have a small condensing boiler inside to provide the extra heat requirement. This has the disadvantage of not totally reducing our dependency on natural gas. Sterling engines have not sold well despite their FIT support.
How do batteries compare in terms of overall efficiency?
Figures are hard to come by but it looks like for lithium cells the efficiency of both charging and discharging is about 90%. This would give around an 80% return overall. It should be added that microbial fuel cells (in which I did my doctorate) have an fuel to electricity conversion efficiency of over 90% (this does not include making the fuel though). Microbial fuel cells have their own problems and will only ever be used in niche uses, such as one utilising brewery waste in Australia.
The comparison with batteries does raise an issue though, the hydrogen economy would require a whole new infrastructure. Compare this with electricity which is ubiquitous. Existing pipes could not be used since hydrogen being a smaller less dense molecule would leak and that unconverted by lightening to water would leak into space. This is happening to our stocks of helium (slightly heavier and less dense). Helium is mined (yes you did read this right), and we are depleting our reserves.
Why are people so enthusiastic about the hydrogen economy?
The answer is most are not. A small number of academics are, one could be cynical and say this is about grant money, but its also seen as sexy and futuristic. They may also have concluded that energy in the future is going be such a huge problem it will require desperate measures. Part of the problem is that most only talk about the efficiency of the fuel to electricity. For this compared to a heat engine (e.g. a petrol engine) with its mechanical parts fuels cells are more efficient, however this is only half the process. Its instructive though in terms of transport private companies have decided lithium is the way forward, there are no hydrogen vehicles on the market but all major manufacturers are marketing electric cars. There are however some planning trials of a small number of fuel cell cars.
Neil
Image from openclipart.org.
References
“Fuel Cell Handbook” (Seventh Edition) by EG&G Technical Services, Inc. U.S. Department of Energy. A good general look at fuel cells and a bit on thermodynamics.
“The Future of the Hydrogen Economy: Bright or Bleak?” Version of 15 April 2003 updated for distribution at the 2003 Fuel Cell Seminar 3 – 7 November 2003, Bossel and Eliasson. Looks at the entire chain including calculations on the energy used to compress and transport hydrogen. The authors conclude if we must use fuel cells as storage then it would be better to use it converted to methane. This has higher embodied energy and does not require new infrastructure.
“Principles and problems in Physical Chemistry for Biochemistry for Biochemists“, Dwek and Price. Written by one of my lecturers this covers kinetics, equilibrium and thermodynamics very clearly.
“Microbiology“, Prescott et al., best and simplest explanation of kinetics, equilibrium and thermodynamics I have yet found, also explains biohydrogen as well.
